
News and Updates
Research Highlights
-
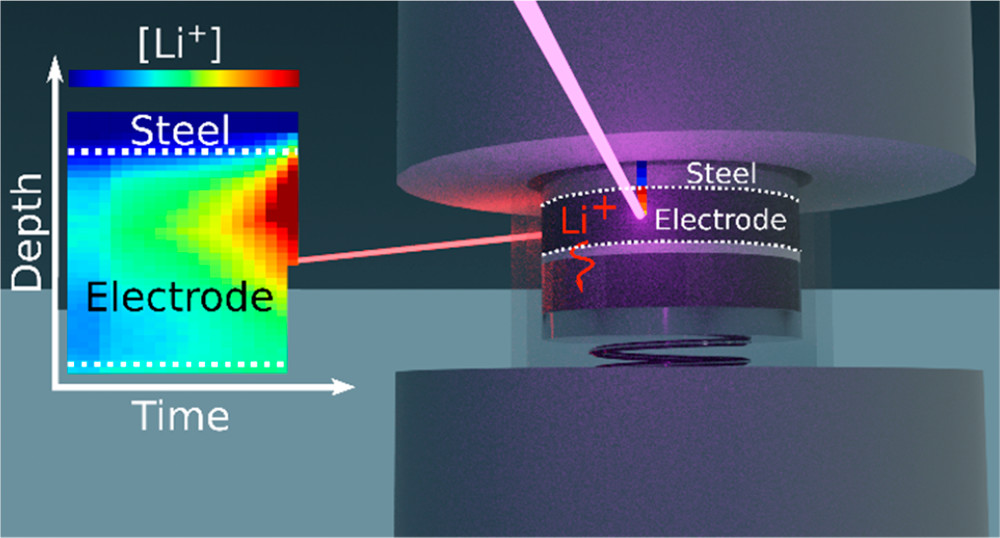
Posted on October 09, 2020Research Highlight : Tracking of Solution-Phase Concentration Profiles in Li-Ion Battery
The trade-off between energy density and power capabilities is a challenge for Li-ion battery design as it highly depends on the complex porous structures that holds the liquid electrolyte. Specifically, mass-transport limitations lead to large concentration gradients in the solution-phase and subsequently to crippling overpotentials. The direct study of these solution-phase concentration profiles in Li-ion battery positive electrodes has been elusive, in part because they are shielded by an opaque and paramagnetic matrix. Herein we present a new methodology employing synchrotron hard X-ray fluorescence to observe the concentration gradient formation within Li-ion battery electrodes in operando. This methodology is substantiated with data collected on a model LiFePO4/Li cell using a 1 M LiAsF6 in 1:1 ethylene carbonate/dimethyl carbonate (EC/DMC) electrolyte under galvanostatic and intermittent charge profiles. As such, the technique holds great promise for optimization of new composite electrodes and for numerical model validation.
-
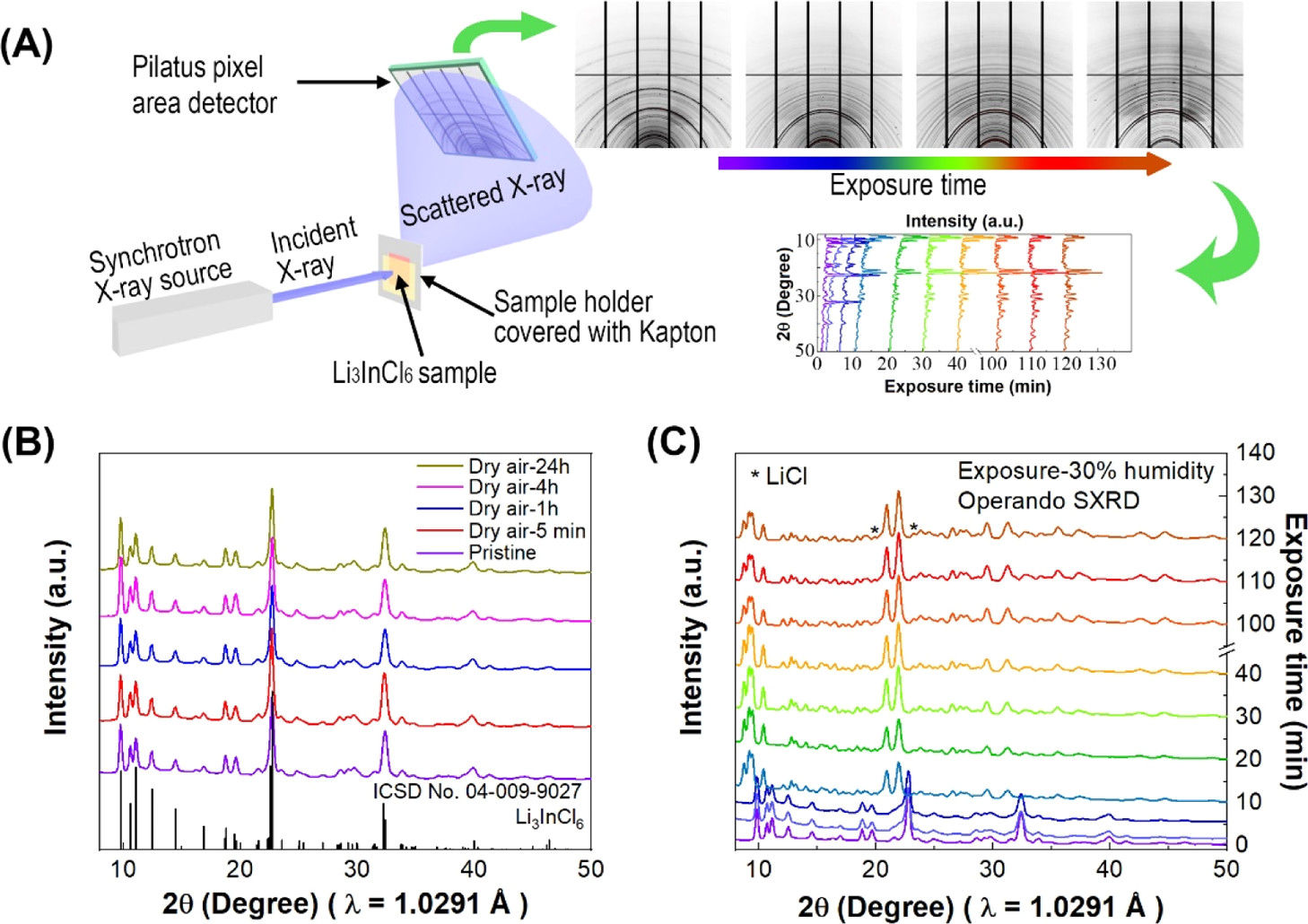
Posted on October 09, 2020Research Highlight : Origin of Moisture Stability of Halide Solid-State Electrolytes
Recently, halide solid-state electrolytes (SSEs) have been reported to exhibit high ionic conductivity and good compatibility with cathode materials. However, the air stability of halide-based electrolytes is one important factor related to ionic conductivity upon exposure to air for practical applications. The instability mechanism of Li3InCl6 toward air is not clearly understood. Herein, we for the first time report the application of operando optical microscopy, Raman spectroscopy, synchrotron-based X-ray powder diffraction, and in situ X-ray absorption near-edge structure for the study of halide electrolyte air stability. Using these methods, we have been able to track the degradation process of Li3InCl6 exposed to air. It is for the first time found that Li3InCl6 is hydrophilic in character, leading to the absorption of moisture from the air, and a portion of Li3InCl6 reacts with absorbed H2O to form In2O3, LiCl, and HCl. Moreover, the remaining electrolyte absorbs H2O to form a hydrate, Li3InCl6·xH2O. The reaction results in a decrease of ionic conductivity. Additionally, the influence of air stability on the practical application of Li3InCl6 has been explored. Li3InCl6 shows much better stability against air with low humidity (3%) and in battery dry rooms, making it a promising SSE for application in the commercial lithium-ion manufacturing industry.
-
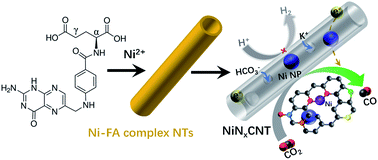
Posted on February 25, 2020Research Highlight : An efficient electrocatalyst for CO2 reduction
Electrochemical reduction of CO2 to value-added products with high selectivity has attracted significant research interest. The development of earth-abundant and low-cost electrocatalysts is the key in this process. Herein, an efficient CO2 reduction electrocatalyst, comprising Ni and N in situ co-doped into porous and Ni nanoparticle-embedded carbon nanotubes (NiNxCNT), is developed from a sustainable and representative bioligand – folic acid. The synthesis process is straightforward, with the crucial step being the chelation of folic acid and Ni ions into uniform tubular metal–organic complex precursor. The resulted NiNxCNT catalyst exhibits a CO partial current density of 9.0 mA cm−2 at −0.676 V versus RHE and a high selectivity towards CO (>98%) in a wide potential range of −0.676 to −0.976 V versus RHE. Furthermore, the electrode shows little current decay over a period of total 44 h continuous operation at different potentials. The notable performance here is attributed to the synergistic effect of rich Ni–Nx sites and hierarchically porous nanotube structure. The findings of this study will open new avenues for developing inexpensive and high-performance CNT-based electrocatalysts for CO2 utilization.
-
Posted on February 25, 2020Research Highlight : VO2 microrod from the reduction of V2O5 thin films
VO2 microrods (MRs) have been successfully synthesized by the reduction of V2O5 thin films. The effects of reduction temperature and thickness of the precursor V2O5 thin films on the stochiometry and morphology of the resulting VO2 MRs have been studied in this work. Results show that size of the VO2 MRs increases with increasing precursor thickness due to the enlargement of the droplets during the melting of V2O5 thin films. Further, the growth of VO2 MRs from V2O5 droplets on a sapphire substrate form a unique angle between each other, which is probably due to epitaxial growth. Reduction temperatures between 600 °C and 650 °C are preferred for the growth of VO2 MRs, while temperatures lower or higher than this range produce V2O5 nanostructures or VO2 nanoparticles, respectively. HRTEM shows that during VO2 MR’s growth, the (110) plane remains parallel to the Si substrate, and a 10 nm amorphous interface was formed between the VO2 MR and the Si substrate. Optical properties of the MRs were examined by carrying out light scattering experiments as a function of temperature to identify the temperature range of insulator-to-metal (IM) and metal-to-insulator (MI) transition characteristics. The optical scattering experiments show the large influence of microrod (MR) size and distribution on the characteristics of MI and IM transitions. The transition behavior can range from polycrystalline film type transition to new unique features.
-
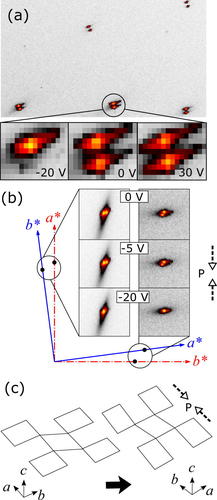
Posted on August 17, 2018Research Highlight : Twin domains in La2−xSrxCuO4 under uniaxial pressure
A study of twin domains in La2−xSrxCuO4 under uniaxial pressure is reported. Using bulk-sensitive X-ray microdiffraction in Laue geometry, the distribution of twin domains at room temperature is imaged. When compressive uniaxial pressure is applied along one of the in-plane crystallographic axes, the domain population changes dramatically. It is observed that the twin domain with a shorter lattice parameter along the direction of pressure is unstable under compression and disappears completely only under moderate pressure. On the other hand, application of tensile pressure changes the domain structure only slightly, demonstrating the asymmetric response of the sample to uniaxial pressure. The observations suggest that a crystal's response to uniaxial pressure is complex and could deviate easily from the linear-response regime.
-
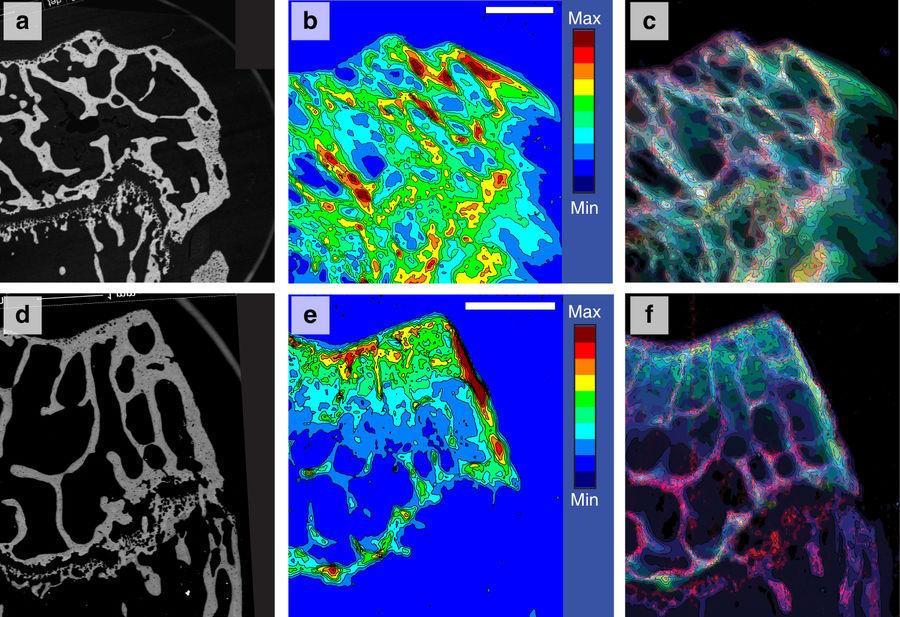
Posted on March 09, 2018Research Highlight : Accumulation of persistent tungsten in bone
Tungsten accumulates in bone but is neither labile nor inert once absorbed. Tungsten’s relatively high cytosolic solubility and availability are problematic given its association with childhood lymphocytic leukemia. In light of tungsten’s technological prevalence, and the increased concern of regulatory agencies, the chemical form and localization in mice exposed to tungsten through drinking water is characterized here. Using X-ray fluorescence spectroscopy, accumulation of tungsten in bone tissue is reported with some sites having ~10-fold greater intensities than background levels. The long bone tissue studied includes cortical, cancellous and bone marrow. Persistence of tungsten in cortical bone tissue following removal of the source indicates that it is retained in an insoluble form. The X-ray absorption near-edge structure spectra for tungsten in these tissues indicate that it is no longer in the originally administered form, orthotungstate, but rather resembles the heteropolytungsate species, phosphotungstate.
-

Posted on March 09, 2018Research Highlight : Formation of conductive phase in LFP during carbon coating
Carbon coating is a commonly employed technique for improving the conductivity of active materials in lithium ion batteries. The carbon coating process involves pyrolysis of organic substance on lithium iron phosphate particles at elevated temperature to create a highly reducing atmosphere. This may trigger the formation of secondary phases in the active materials. Here, a conductive phase is observed during the carbon coating process of lithium iron phosphate and the phase content is size, temperature, and annealing atmosphere dependent. The formation of this phase is related to the reducing capability of the carbon coating process. This finding can provide a guidance to control the phase composition of carbon-coated lithium iron phosphate and to tune its quality during the manufacturing process.
-
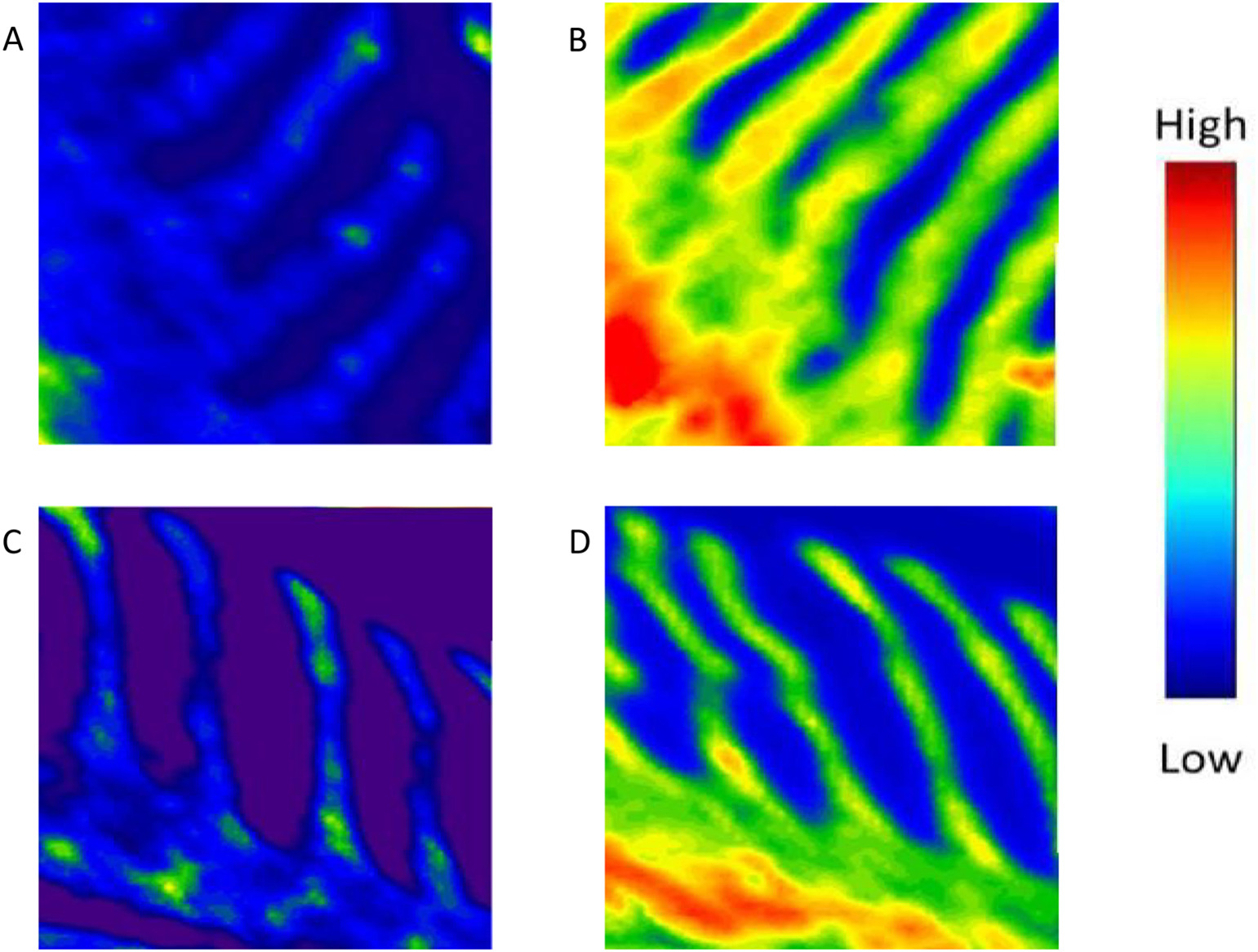
Posted on March 09, 2018Research Highlight : Zinc in Fish Gills
Micro X-ray fluorescence imaging (μ-XFI) and micro X-ray absorption near-edge spectroscopy (μ-XANES) techniques are utilized to investigate Zn distribution profile, its co-localization patterns with Ca, S, and Fe and speciation in the gills of rainbow trout (RBT). Fish (~100 g) were exposed to acutely toxic levels of waterborne Zn alone and in combination with waterborne Cd or Cu for 24 h (each at 1 × 96 h LC50). Gill sections were prepared and analyzed at the VESPERS beamline of the Canadian Light Source. The primary lamellae of the fish gill were found to be the primary area of Zn accumulation. These regions also correspond to the zones of mitochondria rich cells localization in fish gills, supporting the putative roles of these cells in metal uptake. Zn was also found to predominantly co-localize with Ca and S, but not with Fe, indicating the roles of Ca and S in intracellular Zn handling. Zn distribution in the gill was markedly reduced during co-exposure to Cd, but not to Cu, suggesting a competitive interaction between Zn and Cd for uptake. The speciation of Zn in the gill was dominated by Zn-phosphate, Zn-histidine and Zn-cysteine species; however, the interactions of Zn with Cd or Cu resulted in the loss of Zn-cysteine. Overall, our findings provide important novel insights into the interactions of Zn, Cd and Cu in the fish gill, which may ultimately help to explain the mechanisms underlying the acute toxicity of these metals in binary mixture to fish.Comparative Biochemistry and Physiology Part C, 206-207, 23-31(2018)
-
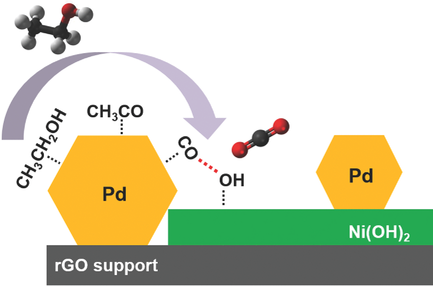
Posted on August 01, 2017Research Highlight : Promoting Effect of Ni(OH)2 on Palladium Nanocrystals
Most electrocatalysts for the ethanol oxidation reaction suffer from extremely limited operational durability and poor selectivity toward the CC bond cleavage. In spite of tremendous efforts over the past several decades, little progress has been made in this regard. This study reports the remarkable promoting effect of Ni(OH)2 on Pd nanocrystals for electrocatalytic ethanol oxidation reaction in alkaline solution. A hybrid electrocatalyst consisting of intimately mixed nanosized Pd particles, defective Ni(OH)2 nanoflakes, and a graphene support is prepared via a two-step solution method. The optimal product exhibits a high mass-specific peak current of >1500 mA mg−1Pd, and excellent operational durability forms both cycling and chronoamperometric measurements in alkaline solution. Most impressively, this hybrid catalyst retains a mass-specific current of 440 mA mg−1 even after 20 000 s of chronoamperometric testing, and its original activity can be regenerated via simple cyclic voltammetry cycles in clean KOH. This great catalyst durability is understood based on both CO stripping and in situ attenuated total reflection infrared experiments suggesting that the presence of Ni(OH)2 alleviates the poisoning of Pd nanocrystals by carbonaceous intermediates. The incorporation of Ni(OH)2 also markedly shifts the reaction selectivity from the originally predominant C2 pathway toward the more desirable C1 pathway, even at room temperature.
-
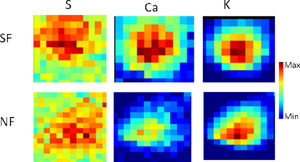
Posted on March 02, 2017Research Highlight : Biochemical Alteration in Cancer-Associated Fibroblasts
The microenvironment of a tumor changes chemically and morphologically during cancer progression. Cancer-stimulated fibroblasts promote tumor growth, however, the mechanism of the transition to a cancer-stimulated fibroblast remains elusive. Here, the multi-modal spectroscopic methods Fourier transform infrared imaging (FTIRI), X-ray absorption spectroscopy (XAS) and X-ray fluorescence imaging (XFI) are used to characterize molecular and atomic alterations that occur in cancer-stimulated fibroblasts. In addition to chemical changes in lipids (olefinic and acyl chain) and protein aggregation observed with FTIRI, a new infrared biomarker for oxidative stress in stimulated fibroblasts is reported. Oxidative stress is observed to cause lipid peroxidation, which leads to the appearance of a new band at 1721 cm−1, assigned to 4-hydroxynonenal. Complementary to FTIRI, XFI is well suited to determining atom concentrations and XAS can reveal the speciation of individual elements. XFI reveals increased concentrations of P, S, K, Ca within stimulated fibroblasts. Furthermore, XAS studies reveal alterations in the speciation of S and Ca in stimulated fibroblasts, which might provide insight into the mechanisms of cancer progression. Using XFI, not only is the concentration change of individual elements observed, but also the subcellular localization. This study demonstrates the wealth of biochemical information provided by a multi-modal imaging approach and highlights new avenues for future research into the microenvironment of breast tumors.
-

Posted on February 13, 2017Research Highlight : Chemical Biology in the Embryo
Proteoglycans (PGs) are heavily glycosylated proteins that play major structural and biological roles in many tissues. Proteoglycans are abundant in cartilage extracellular matrix; their loss is a main feature of the joint disease osteoarthritis. Proteoglycan function is regulated by sulfation–sulfate ester formation with specific sugar residues. Visualization of sulfation within cartilage matrix would yield vital insights into its biological roles. We present synchrotron-based X-ray fluorescence imaging of developing zebrafish cartilage, providing the first in situ maps of sulfate ester distribution. Levels of both sulfur and sulfate esters decrease as cartilage develops through late phase differentiation (maturation or hypertrophy), suggesting a functional link between cartilage matrix sulfur content and chondrocyte differentiation. Genetic experiments confirm that sulfate ester levels were due to cartilage proteoglycans and support the hypothesis that sulfate ester levels regulate chondrocyte differentiation. Surprisingly, in the PG synthesis mutant, the total level of sulfur was not significantly reduced, suggesting sulfur is distributed in an alternative chemical form during lowered cartilage proteoglycan production. Fourier transform infrared imaging indicated increased levels of protein in the mutant fish, suggesting that this alternative sulfur form might be ascribed to an increased level of protein synthesis in the mutant fish, as part of a compensatory mechanism.
-
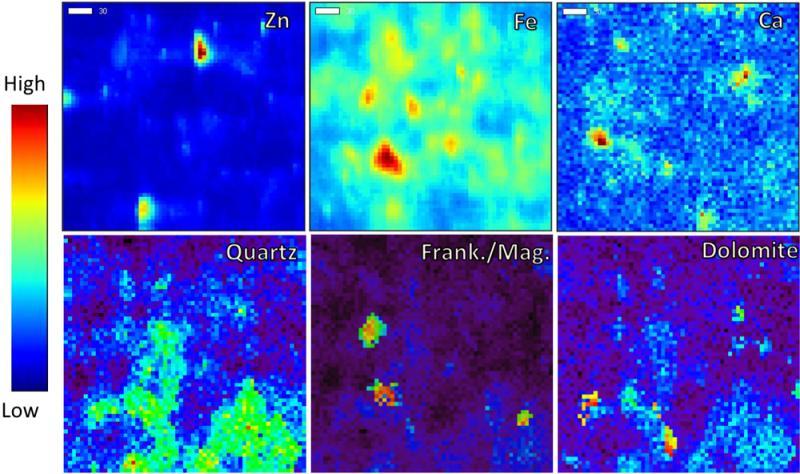
Posted on February 13, 2017Research Highlight : Zinc Speciation in Soils
Anthropogenic activities at the HudBay Minerals, Inc., Flin Flon (Manitoba, Canada) mining and processing facility have severely affected the surrounding boreal forest ecosystem. Soil contamination occurred via a combination of metal and sulfuric acid deposition and has resulted in forest dieback and ineffective natural recovery. A community-led effort to revegetate areas of the landscape through the application of a dolomitic limestone has been met with varied success. Zinc (Zn) speciation has shown to be closely linked to the presence or absence of an invasive metal-tolerant grass species, with soils being broadly classed into two revegetation response groups. Group I, characterized by the absence of metal-tolerant grasses, and group II, characterized by the presence of metal-tolerant grasses. The systematic approach used to lime areas of the landscape produced a liming chronosequence for each group. This study used a combination of X-ray absorption spectroscopy, X-ray fluorescence mapping, and X-ray diffraction techniques to determine the effect of liming on Zn speciation in these chronosequences. Liming group I soils resulted in the formation of a neo-phase Zn-Al-hydroxy interlayer coprecipitate and subsequent rapid boreal forest revegetation. The effect of liming on Zn speciation on the group II soils resulted in a gradual transition of increasingly stable adsorption species, culminating with a stable Zn-Al-layered double hydroxide precipitate. Boreal forest vegetation has failed to recolonize group II soils during the study. However, the formation of the layered double hydroxide species resulted in a significant reduction in CaCl2–extractable Zn. Further research is required to determine how to promote the revegetation of these soils.
-
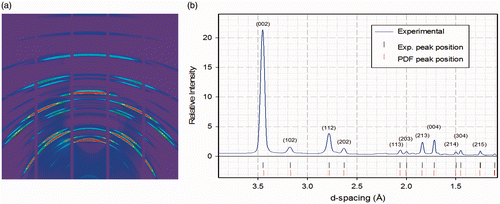
Posted on February 13, 2017Research Highlight : Tooth of a Dinosaur
Synchrotron micro X-ray fluorescence (XRF) spectroscopy with two-dimensional element mapping, micro X-ray diffraction (XRD), electron spin resonance spectroscopy (ESR) and atomic force microscopy (AFM) were used to investigate the chemical and structural nature of the enamel of a tooth from Troodon, a small theropod dinosaur. These methods show that the crystallites in the Troodon tooth are submicron-sized carbonated calcium hydroxyapatite, which are semi-randomly oriented with a preferred orientation of (002) towards the surface of the tooth. Transition metal ions are distributed in the voids between crystallite clusters. Comparison of the ESR spectra indicates that the Troodon tooth had less exposure to UV than a fossilized crocodile tooth.
-
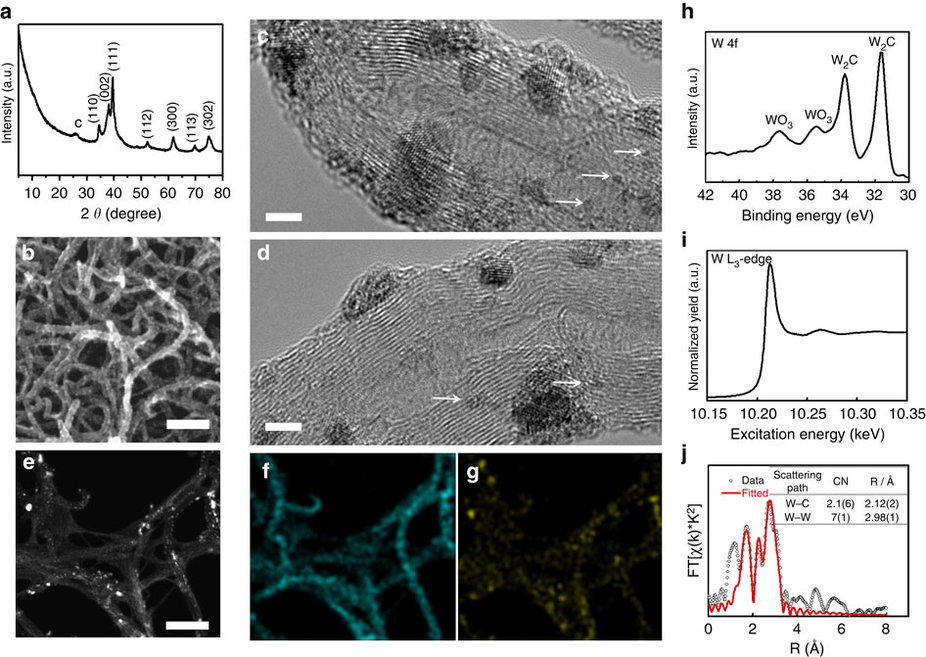
Posted on February 13, 2017Research Highlight : Ultrasmall W2C Nanoparticle for HER
Earlier research has been primarily focused on WC as one of the most promising earth-abundant electrocatalysts for hydrogen evolution reaction (HER), whereas the other compound in this carbide family—W2C—has received far less attention. Our theoretical calculations suggest that such a focus is misplaced and W2C is potentially more HER-active than WC. Nevertheless, the preparation of phase pure and sintering-free W2C nanostructures represents a formidable challenge. Here we develop an improved carburization method and successfully prepare ultrasmall and phase-pure W2C nanoparticles. When evaluated for HER electrocatalysis, W2C nanoparticles exhibit a small onset overpotential of 50 mV, a Tafel slope of 45 mV dec−1 and outstanding long-term cycling stability, which are dramatically improved over all existing WC-based materials. In addition, the integration of W2C nanoparticles with p-type Si nanowires enables highly active and sustainable solar-driven hydrogen production. Our results highlight the great potential of this traditionally non-popular material in HER electrocatalysis.